While we can all agree that single-cell sequencing is a powerful tool in and of itself, it misses out on a critical aspect of biology – the positional context of cells relative to one another. Most tissues in the body are not simply made up of one type of cell repeated over and over, but instead comprise a wide variety of different cell types arranged in specific patterns. And even individual cells of the same type will adopt different roles depending on where they are located, in the same way that individual humans will act differently in a city than they would in a suburb or rural area. Each cell type supports neighboring cells, and they in turn rely on communication and feedback from their neighbors. Distinct sub-structures within a tissue must be properly arranged and maintained; deviation may result in, or be an underlying cause of, dysfunction or disease progression. This concept of uncovering cell behaviors, also known as “Cellular Neighborhood Analysis” underscores the need to understand the complex interactions in tissue biology.
Unfortunately, earlier methods of genomic analysis required breaking down the intact tissue in order to access and sort individual cells, so there was no way to comprehensively characterize a cell while also knowing its exact arrangement within the tissue.
This is where the “spatial” part of spatial genomics comes in. With the development of technologies such as MERFISH technology, scientists are able to simultaneously profile a multitude of cells on a transcriptomic level (i.e. which genes are expressed in each cell) while retaining the spatial context of the intact tissue. The implications of this combined analysis are particularly interesting in tissues such as the brain, with its complex web of interactions between different neuronal sub-types, or the tumor microenvironment, which can alter to evade the immune response or therapy.
What are the keys to successful spatial genomics profiling?
There are three factors to consider when assessing the quality of spatial genomics data: Accuracy, sensitivity and resolution.
Accuracy & Specificity: This is the ability to measure exactly what you want to measure, and not mistake it for something else. Accuracy is often traded off when looking at multiple targets simultaneously when trying to correctly and comprehensively characterize multiple different cell sub-types within a single tissue. Advances in technology, such as Vizgen’s MERFISH platform, use a multiplexed barcoding scheme to increase the number of genes assessed. This barcoding system is designed with error robustness; the barcode sequences are designed to tolerate a certain number of misread elements before one barcode is incorrectly identified as another. If there is a readout error – caused by excessive background fluorescence, for example – the system can assign the readout to the nearest correct barcode or the error can be detected and the corresponding transcript discarded, ensuring an accurate measurement.
Sensitivity: Some genes are expressed at varying levels compared to others, but just because a gene is expressed at a low level, it doesn’t mean that the product of that gene isn’t part of a critical biological process. For effective characterization of cells, the capability to measure these genes is essential. Higher sensitivity means increased ability to detect genes across a wide dynamic range, from the high to low expressors.
Resolution: This is the ability to discriminate between two objects that are close together. For spatial genomics, where you are trying to analyze the expression levels of multiple genes as well as their localization within a tissue, resolution is very important. With cutting edge technologies such as MERSCOPE, scientists are able to localize transcripts at a subcellular level, from whole tissue section to single-cell and finally sub-cellular imaging across the whole tissue, with ≤100 nm resolution. To put that into context, 100 nm is about 100x smaller than a human T cell.
Compared to other options such as slide-seq or array-based sequence methods, MERFISH offers the highest detection efficiency and resolution for spatially profiling the transcriptome. A paper published in Nature Reviews, Genetics in July of this year describes the developing landscape of spatial genomic technologies and compares the different approaches. With over 20 peer-reviewed studies published since 2015, MERFISH has already made its mark on the scientific community – but this is just the beginning
While getting started with spatial genomics can feel daunting, researchers now have access to an all-in-one platform solution that provides everything a lab needs to begin running spatial experiments. MERSCOPE is Vizgen’s complete technology solution that integrates the power and potential of MERFISH technology with high-resolution imaging, fluidics, image processing, and automation to help scientists explore spatial context with minimal challenges. Because scientists can design custom gene panels for MERSCOPE experiments, this platform is incredibly versatile across applications and fields of study. In addition to the instrument, reagents, and consumables you need to run advanced spatial genomics experiments, the MERSCOPE Platform includes intuitive software that provides support for spatial data visualization and analysis.
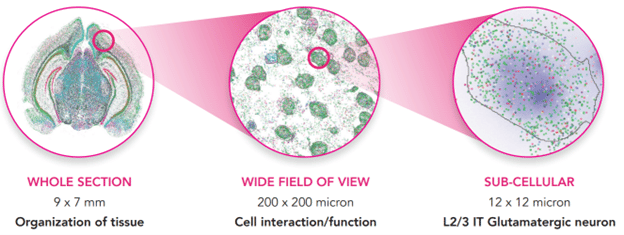
The MERSCOPE Platform brings your spatial genomics workflow from tissue sample to sub-cellular levels, allowing you to uncover tissue organization, cell function and interactions, and gene expression within individual cells
In the words of Vizgen President and CEO Terry Lo:
“Single-cell spatial genomics is a true game-changer in how we are able to understand complex biological systems and it is incredibly gratifying to see how the research community is embracing our ground-breaking MERSCOPE platform for unparalleled genomics insights, accelerating biological research and discovery.”